Case Report / Open Access
DOI: 10.31488/bjcr.207
Efficacy of the Novel Regimen Pemetrexed-Zanubrutinib in a Patient with Chronic Lymphocytic Leukemia and Central Nervous System Diffuse Large B Cell Lymphoma Presenting as Richter’s Transformations Transformation
Julia Steger1, Jeremy Purow1, Tiba Al Sagheer1,2, Ariel Perez Perez1,2 , Yazmin Odia , MD1,2, Zhijian Chen1, 2, Claudia Torralbas1, Arun Maharaj1, Manmeet Ahluwalia1, 2, Yuliya Linhares, MD*,1,2
1. Florida International University, Herbert Wertheim College of Medicine- 11200 SW 8th St AHC2, Miami, FL 331992
2. Miami Cancer Institute, Baptist Health South Florida- 8900 N Kendall Drive, Miami, FL 33176
*Corresponding author:Julia Steger, Florida International University, Herbert Wertheim College of Medicine- 11200 SW 8th St AHC2, Miami, FL 33199, YuliyaL@baptisthealth.net
Abstract
Richter’s transformation (RT) is the transformation of chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL) into an aggressive lymphoma, most commonly diffuse large B-cell lymphoma (DLBCL). RT presenting exclusively in the central nervous system (CNS-RT) is exceedingly rare, and no standardized treatment protocols currently exist. High-dose methotrexate (HD-MTX), the cornerstone of treatment for primary CNS lymphoma (PCNSL), has shown limited efficacy in CNS-RT. Pemetrexed, a folate antimetabolite, and zanubrutinib, a Bruton kinase inhibitor (BTKi), have both demonstrated activity in relapsed or refractory (R/R) CNS lymphoma, but their activity in CNS-RT has not been established. We report a case of a 60-year-old male with CLL who developed isolated, clonally related CNS-RT, refractory to HD-MTX. The patient was treated with Pemetrexed-Zanubrutinib (Pem-Zan) per the NCT05681195 protocol and achieved complete remission (CR) following four cycles of therapy. The patient underwent thiotepa-carmustine conditioning and autologous stem cell transplant (ASCT) consolidation, and remains in remission from CNS-RT. This case illustrates the efficacy of Pem-Zan in a patient with CNS-RT. The NCT05681195 protocol which examines the efficacy in Pem-Zan in relapsed or refractory PCNSL and secondary CNS lymphoma is ongoing.
Key Words: Richter’s Transformation, Pemetrexed, Zanubrutinib, Primary CNS Lymphoma
Introduction
Richter’s transformation (RT) is the transformation of chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL) into a more aggressive lymphoma, most commonly diffuse large B-cell lymphoma (DLBCL) [1-3] Transformation into Hodgkin lymphoma and other rare subtypes has been reported [4]. This complication affects up to 15% of CLL patients throughout their disease, with an annual incidence of approximately 0.5-1% [1-3, 5]. The median time from CLL diagnosis to RT is approximately 4 years, even though some patients present with concomitant transformation. The median age at diagnosis is approximately 66 years [6]. Certain CLL features are associated with an increased risk of RT, such as advanced-stage disease, unmutated Immunoglobulin Heavy Chain Variable (IGHV) status, the IGHV stereotyped subset number 8 (IGHV4-39-IGHJ5), neurogenic locus notch homolog protein 1 (NOTCH1) mutations, deletion 17p and/or TP53 mutation, and del (11q). Near tetraploidy is associated with a high risk of RT in patients receiving ibrutinib [5, 7]. In a large study, deletion 13q had a protective effect on the risk of RT [8]. RT is associated with rapid progression and poor prognosis, especially if the transformation is clonally related [9, 10]. Clonally unrelated RT patients have a better prognosis with a median progression-free survival (PFS) of 62.5 months versus 14.2 months in those with clonally related disease [9]. The cell of origin is generally of an activated B-cell (ABC) type, expressing post-germinal center markers such as IRF-4, whereas only 5–10% display a germinal center B-cell (GCB) phenotype, expressing CD10 and/or BCL6 [11]. Moreover, CLL markers such as CD5 and CD23 can be dim or negative in RT [2].
While RT predominantly involves lymph nodes, extranodal presentations, including central nervous system (CNS) involvement, have been reported. Isolated CNS involvement in RT is rare, and there are no current standardized treatment protocols [12-15]. Management strategies are generally extrapolated from treatments for primary CNS lymphoma (PCNSL), often including high-dose methotrexate (HD-MTX) and other CNS-penetrant chemotherapeutic agents [13, 16, 17]. Patients with isolated CNS-RT sensitive to HD-MTX can achieve long-term remission with autologous stem cell transplant (ASCT) consolidation [6, 13]. In the current case report, we describe a rare, unique patient who has concurrent CLL with bone marrow and lymph node involvement and isolated HD-MTX refractory clonally related CNS-RT, without evidence of systemic transformation.
Case presentation
A 60-year-old male with a history of CLL not meeting treatment criteria since diagnosis presented with progressive neurological symptoms, including left-sided paralysis, vision impairment, and cognitive decline. Small, palpable axillary lymph nodes were present on the physical exam. Magnetic resonance imaging (MRI) of the brain with and without (w/wo) contrast revealed a large, enhancing right parietal brain lesion that was 6.0 x 7.6 x 6.1 cm with significant mass effect and a 5 mm right-to-left midline shift and signs of early ventricular entrapment (Figure 1). Complete blood count demonstrated white blood cell (WBC) count of 44 103/uL with an absolute lymphocytosis at 65%, hemoglobin (Hgb) of 14.1 g/dL, and platelet count of 300 103/uL. Computed tomography (CT) of the chest, abdomen, and pelvis revealed innumerable prominent lymph nodes with borderline increase by size criteria. Needle biopsy of a right axillary lymph node demonstrated complete effacement of the lymph node by small monotonous lymphocytes with irregular nuclear contours and multiple large proliferation centers. The immune phenotype was cluster of differentiation (CD)20 (+), paired box 5 (PAX5) (+), CD5 (+), CD23 (+), CD3 (–), BCL-1 (–), BCL-2 (–), and the Ki67 index was 70% in the proliferation centers. Flow cytometry demonstrated lambda-restriction. Overall morphologic findings were consistent with a histologically aggressive small lymphocytic leukemia/chronic lymphocytic leukemia (SLL/CLL). Karyotype was normal male (46, XY [20]). CLL Fluorescence In Situ Hybridization (FISH) analysis showed chromosome 13q homozygous deletion leading to the deletion in lymphocytic leukemia 1 (DLEU1) and 2 (D138319 region) in 77.3% of the cells (NeoTYPE CLL FISH Panel). Immunoglobulin heavy chain variable region (IGHV) was mutated. Myeloid differentiation primary response 88 (MYD88) leucine position 265 proline (L265P) gain-of-function (GOF) mutation was present while TP53 was wild type (NeoGenomics, NeoTYPE Analysis CLL profile). The lymph node biopsy sample was sent for Tumor Portrait assay with BostonGene. Ribonucleic acid (RNA) sequencing revealed Cyclin D1 expression. Additionally, the bone marrow biopsy revealed 10% involvement of the marrow cellularity by CLL with identical chromosomal abnormality and mutational profile as the lymph node. At the time of presentation, the patient had Binet stage B (intermediate risk) and Rai stage 1 CLL [18, 19].
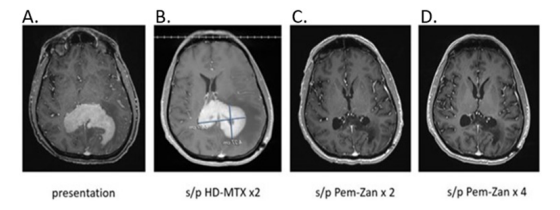
Figure 1:Brain MRI W/WO contrast of a 60-year-old male with PCNSL. Figure 1A was obtained at presentation; Figure 1B shows persistent PCNSL following 2 cycles of HD-MTX. The final two images reveal radiographic improvement after 2 (Figure 1C) and 4 (Figure 1D) cycles of Pem-Zan, respectively.
A right parietal craniotomy was performed, and the pathology report from the removed tumor revealed a diagnosis of LBCL, of a non-germinal center B-cell immunophenotype by Hans’ algorithm [20]. The immune phenotype was CD20 (+), CD5 (+), BCL6 (+), BCL-2 (+), PAX5 (+), multiple myeloma oncogene 1 (MUM–1) (+), MYC protein (+), BCL1(–), CD10 (–), CD138 (–), CD23 (–), CD30 (–), with a Ki–67 index of 90%. No low-grade lymphoma/CLL component was found in the biopsy. Flow cytometry of the tissue showed a CD5 (+) monoclonal B-cell population, kappa-restricted, with bright CD20 expression, CD38 expression, and negative CD23 expression. The LBCL was positive for the genetic mutations CREBBP Y1433_D1435delinsDLE (variant allele frequency, VAF 34.3%), ETV6 S257Tfs9 (VAF 48.4%), and PRDM1 E153 (VAF 45.2%) mutations (NeoGenomics). Further CNS tumor tissue analysis utilizing BostonGene Tumor Portrait assay revealed CD79B Y196N GOF mutation (VAF 100%), PRDM1 E153 loss-of-function mutation (VAF 45.1%), and negativity for MYD88 mutations. LBCL was positive for HLA Class I loss of heterozygosity. Lymphoma genetic classification by next-generation sequencing was consistent with the MCD/A53 subtype. Based on RNA sequencing results, the tumor microenvironment was immune-inflamed with high PDL1 expression (BostonGene). IGHV sequencing of the framework 1 (FR1) clonal rearrangement sequence 1 and FR2 rearrangement 1 in the brain lesion sample had 100% identity with each other, and FR1 and FR2 clonal rearrangements 1 and 2 were detected in the peripheral blood, demonstrating clonal relatedness of the CLL and CNS large B cell lymphoma. This indicates Richter’s transformation in the CNS, albeit with a different light chain expression.
The patient received an initial cycle of HD-MTX 3.5g/m2 with a brief steroid taper with improvement in neurological symptoms. A brain CT scan wo contrast, 12 days post first dose of HD-MTX, demonstrated improvement in mass effect and a 3.1 cm right parietal lesion with surrounding edema consistent with treatment response. The patient then received a second dose of HD-MTX 3.5 g/m2 with a brief dexamethasone taper, which was completed approximately 1 week after HD-MTX administration . However, before the scheduled third dose of MTX, the patient presented with recurrent significant worsening neurologic deficits, including the inability to ambulate. A brain MRI w/wo contrast demonstrated progression of the right parietal lobe mass, now measuring 6.5 x 5.0 x 4.2 cm with confluent periventricular and corpus callosum enhancement of the splenium and body, as well as left parietal lobe involvement (Figure 1). Surrounding T2/FLAIR hyperintensity was observed, which was suggestive of edema and infiltrating tumor. A 7 mm midline shift to the left was noted, but there was no evidence of hydrocephalus. In comparison to prior non-contrast CT scans, the mass effect of the CNS lymphoma was significantly more prominent, with invasion into the posterior horns of both lateral ventricles, causing a midline shift and increased surrounding vasogenic edema. Despite the initial radiological and clinical response to HD-MTX, the patient's disease progressed after two cycles. The patient was started on dexamethasone 4 mg orally 4 times a day with incomplete improvement of neurological symptoms.
The patient was enrolled in a clinical trial of pemetrexed-zanubrutinib (Pem-Zan) (NCT05681195). At that time, the patient had been on 16 mg dexamethasone per day for over 9 days. A CT scan with contrast of neck, chest, abdomen, and pelvis, testicular Doppler US, and a full spinal MRI w/wo contrast found no new systemic involvement, and stable small size lymphadenopathy consistent with a history of CLL. No evidence of lymphoma was found on ophthalmologic examination. The patient was then treated with pemetrexed 900 mg/m2 intravenously every 3 weeks for a total of 5 cycles and zanubrutinib 160 orally twice per day. Per the protocol, zanubrutinib was held two days before, on the day of, and on the day after pemetrexed to avoid drug to drug interaction (DDI). The rationale for this measure is based on report by Lionakis et al. 2017 indicating that there may be an antagonistic relationship between BTK inhibitors and anti-folate agents [21]. Supportive care consisted of peg-filgrastim with each cycle, folic acid and B12 supplementation, and dexamethasone the day before, the day of, and the day after pemetrexed (Pemetrexed Package Insert). Following two cycles of Pem-Zan, an MRI of the brain w/wo contrast revealed near complete resolution of extensive areas of confluent periventricular enhancement and corpus callosum enhancement, with minimal residual corpus callosum splenium linear enhancement and reduction in vasogenic edema and mass effect (Figure 1). This was consistent with a partial response (PR) in accordance with the International PCNSL Collaborative Group's (IPCG) criteria [22]. Dexamethasone was gradually tapered and discontinued completely on the day 9 of cycle 3 of Pem-Zan.
Upon the completion of cycle 4 of Pem-Zan, an MRI of the brain wo contrast revealed continued treatment response as indicated by complete resolution of enhancement in the treated tumor area, reduced residual FLAIR signal, and some cystic changes consistent with unconfirmed CR (CRu) [22] (Figure 1). The patient reported feeling well overall and had intact cognitive function (mini-mental state examination, MME 29/30). The patient completed a total of 5 cycles of Pem-Zan treatment without significant complications, with resolution of neurologic symptoms. The patient experienced transient grade 1 thrombocytopenia, which resolved spontaneously, as well as transient grade 1 elevation in liver function tests, which resolved with dietary changes. There were no grade 2 or higher adverse events reported. The patient remained an outpatient throughout Pem-Zan treatment. Before ASCT, positron emission tomography (PET) CT showed no metabolically active/high-grade lymphoma (Deauville score of 1). Bone marrow biopsy demonstrated 1% involvement by CLL and was negative for LBCL. The patient underwent carmustine-thiotepa 20 mg/kg (BCNU-TT 20) conditioning, ASCT, and experienced neutrophil engraftment on day +9. They were discharged home on day +13, remaining transfusion-free [23].
Brain MRI w/wo contrast continues to demonstrate CNS-RT remission 10 months post-ASCT. PET-CT did not show systemic relapse eight months post-ASCT. The ClonoSEQ® Minimal Residual Disease (MRD) assay was positive for the CLL sequence, indicating MRD presence on day +103 (Adaptive). CBC and was monitored for signs of hematological CLL relapse and has remained negative as of day +292.
Discussion
CNS involvement with Richter’s transformation is extremely rare, and its incidence is not defined. Distinct biological risk factors for CNS-RT are not described and are thought to be the same as those for systemic RT. In the largest series of RT from the Mayo Clinic CLL database, about 1 out of 14 RT patients had CNS involvement (7%). Of the 15 RT patients with CNS involvement, 10 had CNS involvement at RT diagnosis; of those, 4 had isolated CNS-RT, while 5 developed CNS involvement with RT progression. The median time from RT diagnosis to CNS involvement was 14.9 months (range 2.8-41.1). Six were of the GCB subtype while 3 were of the non-GCB subtype [13].
Molecular characteristics unique to CNS-RT have not been described. Approximately 96% of PCNSLs carry mutations in the NF-κB signaling network, leading to its aberrant activation. CD79B, which is a part of the BCR complex, is mutated in 83% of cases of PCNSL, while MYD88, an adaptor protein downstream of Toll-like receptor, is mutated in 76% of PCNSL, with 67% of PCNSLs reported to be double mutant [24-26]. Activated Bruton kinase (BTK) preferentially forms a complex with MYD88L265P mutant, leading to the activation of the NF-κB pathway. BTK inhibition results in decreased formation of the MYD88-BTK complex with CD79B and MYD88 double mutants, benefiting the most from BTK inhibition [27, 28]. The case of this patient is unique as the CLL carried MYD88 L265P gain of function (GOF) mutation, while the CNS LBCL lesion was positive for CD79B Y196N GOF mutation. MYD88 mutations are uncommon in CLL and were reported to occur in 3.1% of a large cohort of 1779 patients, where 2.1% of patients had MYD88L265P GOF mutation, while the rest had MYD88 non-L265P mutations. Patient characteristics of MYD88L265P mutants differ from those of MYD88 non-L265P. MYD88L265P mutants have lower beta-2 microglobulin, are more frequently negative for CD38 and ZAP70, have a higher frequency of mutated IGHV and isolated del (13q14.3), and a lower frequency of del(11q22.3) and mutations of NOTCH1 and SF3B1.
MYD88 non-L265P mutants are more similar to CLL patients with wild-type MYD88. There is no difference in time to first treatment when comparing MYD88-mutated vs. wild-type CLL patients before and after stratification according to IGHV mutation status [29]. While MYD88 mutations are associated with the extra-lymphatic disease in DLBCL, and PCNSL is enriched in MYD88 mutations, with various studies showing an average 60.8% (33-100%) of PCNSL carrying the MYD88 L265P mutation, there are no consistent reports of the association of MYD88L265P mutation in CLL with CNS involvement or CNS RT [28]. There is only one case report of a MYD88 mutation in CLL as well as in CNS-RT, large B cell histology, which hints at the possibility of its significance in CNS-RT pathogenesis [17]. Another case report describes a CNS-RT double mutant for MYD88L265P GOF and CD79BY196F GOF mutations responsive to ibrutinib, yet again prompting further exploration of the BCR signaling axis mutations in CNS-RT [30]. It is possible that the relationship between the incidence and pathogenicity of the BCR-signaling axis mutation has not been reported due to the low testing for MYD88 and CD79B mutations in CLL, especially in CNS-RT pathologic samples [17].
The patient in the current case report represents an exceptionally rare case of RT; there was no evidence of transformed disease outside of the CNS [12, 13]. Additionally, the patient had CLL, positive for 13q deletion, which is typically a favorable prognostic factor in CLL and is reported to be protective of RT [31]. Another unusual feature in our patient is a mutated IGHV, as about 80% of RT cases are IGHV-unmutated [32].
Notably, CLL was lambda-restricted, and the RT was kappa-restricted. This raises the question of whether the LBCL lesion in the brain was related to the original CLL.
However, CLL may be biclonal with distinct kappa and lambda chain-restricted B cell populations [33, 34]. A kappa-restricted subclone may have existed within the original CLL and remained undetected. The RS may have evolved from the kappa-restricted subclone that may have gone undetected in the CLL. It is also possible that the B cell clone that evolved into LBCL separated at the pro-B cell stage, the stage at which IGHV rearrangement occurs, as evidenced by IGHV sequencing, which showed clonal relatedness between the CLL and CNS-RT lesion [35]. Clonal relatedness of CLL and RT was reported to be associated with treatment resistance and particularly poor outcomes, with the median OS of 14.2 months versus 62.5 months in the clonally unrelated cohort [4].
In the Mayo retrospective CNS-RT series, four patients with isolated CNS-RT were treated with HD-MTX-based regimens, of whom 3 achieved CR and were consolidated with ASCT, followed by durable remission. The patient who failed to achieve CR with HD-MTX received whole-body radiation therapy (WBRT) at progression and subsequently succumbed to the disease. The six patients with concomitant CNS and systemic RT received CNS-directed (HD MTX, n=5 or WBRT, n=1) and systemic therapy with very short disease control time and limited survival (2.1-19.1 months). All 5 patients who developed CNS involvement at RT progression after 1-2 lines of therapy received HD-MTX-based regimens, albeit with short responses and short survival (0.8-9.2 months). Only 1 patient had CR but eventually relapsed after 2 years (32-month survival). The OS in patients with RT and CNS involvement was 9.4 months. However, those with CNS involvement at initial RT diagnosis had numerically longer OS than those who developed CNS involvement at RT progression (median OS 13 vs 4.1 months). Patients with isolated CNS-RT involvement had better OS compared to patients with both CNS and systemic RT involvement (median OS 32 vs 5.3 months, P = 0.009). None of the patients who did not achieve a CR with HD-MTX-based regimens survived. Only the 3 patients with isolated CNS-RT who achieved CR with HD-MTX and were consolidated with ASCT achieved long-term PFS (77-42 months). These results highlight the dismal prognosis in CNS-RT patients who fail to achieve CR to HD-MTX [13].
Systemic RT is frequently chemotherapy-resistant, with a dismal one-year survival of less than 50% with traditional treatment approaches [36]. Nevertheless, the introduction of targeted therapies, such as checkpoint inhibitors (CPIs) and BTKis, holds promise in improving Richter’s outcomes. Richter’s patients treated with a CPI tislelizumab and zanubrutinib had an OR of 58.3% and a 12-month OS of 74.7% [37]. Among RT patients treated on the MOLTO trial with venetoclax, atezolizumab, and obinutuzumab, 67.9% responded. The median durations of response, PFS, and OS were 11.7, 16.2, and 31.6 months, respectively [38]. Although lisocabtagene maraleucel is currently FDA-approved for CLL, patients with CNS involvement and Richter’s transformation were excluded from the pivotal TRANSCEND CLL 004 trial, limiting the generalizability of these results to this high-risk population [39-41] (NCT04792489). Nevertheless, there is no described standard strategy to treat CNS-RT, especially relapse/refractory (R/R) to HD-MTX regimens.
The current case study patient was initially treated with HD-MTX, with PD signifying a dismal prognosis [13]. Given the lack of standard treatment options, the patient was enrolled in the NCT05681195 protocol and treated with Pem-Zan. The patient achieved a PR after 2 cycles of treatment, and subsequently a CRu after 4 cycles of treatment. After 5 cycles, he underwent carmustine-thiotepa conditioning and ASCT. The patient remains in remission from CNS-RT at 10 months post-ASCT, while CLL was detectable in the blood at MRD level (ClonoSEQ®) at 3.5 months post-ASCT.
Pemetrexed was chosen as a combination partner with zanubrutinib due to its known activity in R/R PCNSL, good safety profile, and outpatient administration. Pemetrexed is a promising antifolate agent that targets multiple steps in the folate production pathway as opposed to MTX, which primarily inhibits dihydrofolate reductase (DHFR). Pemetrexed interrupts thymidylate synthase and DHFR in the purine synthesis pathway and inhibits glycinamide ribonucleotide formyltransferase and aminoimidazole carboxamide formyltransferase in the pyrimidine synthesis pathway. Pemetrexed has demonstrated activity in de novo and HD-MTX refractory PCNSL and secondary CNS lymphoma (SCNSL) [20, 42]. In a study of 12 PCNSL patients, 4 achieved a complete response and 6 had a partial response [20]. Pemetrexed was also studied in a phase I trial for the treatment of R/R CNSL. Main adverse events included fatigue (82.4%), anemia (82.4%), and neutropenia (70.6%). The maximum tolerated dose (MTD) was established at 900 mg/m2. The ORR, including CR+PR, was 57.1%, and the disease control rate (DCR), including PR+CR+SD, was 71.4%. The 6-month PFS and 12-month PFS rates were 35.7% and 21.4%, respectively, with median PFS and OS durations of 4.2 months and 44.5 months, respectively. The estimated 1-year, 2-year, and 5-year survival rates were 78.6%, 64.3%, and 35.7%, respectively. Patients remained on treatment until either a complete response was achieved or cycle eight were completed. From the evaluable patients, the median number of treatment cycles was 6.4 ± 3.4 [43]. In a prospective study, 11 patients with R/R PCNSL were treated with pemetrexed. Ten of 11 patients had failed prior HD-MTX. The median number of doses of pemetrexed administered was 10, with an associated CR rate of 36%, overall response rate of 55% and a disease control rate of 91%. The 6-month PFS was 45%, the median PFS was 5.7 months, and the median OS was 10.1 months. The most common adverse events were hematologic. Only 1 patient required a dose reduction for hematologic toxicity, and 1 patient was unable to restart because of a grade 3 thrombocytopenia [42]. Due to its activity in HD-MTX refractory CNS lymphoma, its favorable toxicity profile, and outpatient administration, pemetrexed can serve as a backbone for the treatment of R/R CNS lymphoma in targeted therapy and immunotherapy trials.
Zanubrutinib is a selective BTK inhibitor with a favorable toxicity profile. It exhibits excellent activity in de novo and R/R CLL [44, 45]. A small study demonstrated that zanubrutinib exhibits excellent blood-brain barrier penetration in humans, supporting its further investigation for the treatment of CNS lymphoma [46]. In a PRiZM+ Phase II platform study (ISRCTN90634455), Zanubrutinib demonstrated durable responses in R/R PCNSL (Fox 2025). Due to the known activity in both CLL and HD-MTX refractory PCNSL, Pem-Zan combination treatment was deemed to be appropriate for a patient with CLL as well as HD-MTX refractory CNS-RT. The patient harbored activating BCR-BTK pathway mutations, MYD88 L265P GOF in CLL, and CD79B Y196N GOF in CNS-RT. Both CLL and CNS-RT responded to the Pem-Zan combination. The CD79B mutation in the CNS-RT lesion may have contributed to its sensitivity to zanubrutinib, highlighting the importance of molecular mutational analysis in the era of targeted therapies. BCNU-TT ASCT was chosen as a consolidative option once a CR was achieved. TT-based ASCT consolidation is the best consolidative therapy in PCNSL, known for achieving durable remissions [47]. Additionally, CNS-RT patients who were consolidated with ASCT can achieve long-term PFS [13].
Conclusion
The Pem-Zan combination therapy was well-tolerated and demonstrated clinical activity in this patient with HD-MTX-refractory CNS-RT. CD79B mutation in the BTK pathway may have contributed to zanubrutinib sensitivity. Pem-Zan is convenient, as it is administered fully on an outpatient basis, unlike most standard CNS-directed regimens. The Pem-Zan (NCT05681195) clinical trial is currently open for enrollment for the relapsed or refractory PCNSL and SCNSL, including large cell CNS transformation of indolent lymphomas.
References
1. Parikh SA, Kay NE, Shanafelt TD. How we treat Richter syndrome. Blood. 2014;123(11):1647–57.
2. Jain P, O’Brien S. Richter’s transformation in chronic lymphocytic leukemia. Oncology (Williston Park). 2012;26(12):1146.
3. Caruso A, Rossi D. Richter syndrome. Curr Oncol Rep. 2021;23(3):29.
4. Tsimberidou AM, O’Brien S, Khouri I, Giles FJ, Kantarjian HM, Champlin R, et al. Clinical outcomes and prognostic factors in patients with Richter’s syndrome treated with chemotherapy or chemoimmunotherapy with or without stem-cell transplantation. J Clin Oncol. 2006;24(15):2343–51.
5. Thompson PA, Siddiqi T. Treatment of Richter’s syndrome. Hematology Am Soc Hematol Educ Program. 2022;2022(1):329–36.
6. Grommes C, Rubenstein JL, DeAngelis LM, Ferreri AJ, Batchelor TT. Comprehensive approach to diagnosis and treatment of newly diagnosed primary CNS lymphoma. Neuro Oncol. 2019;21(3):296–305.
7. Hleuhel MH, Ben-Dali Y, Da Cunha-Bang C, Brieghel C, Clasen-Linde E, Niemann CU, et al. Risk factors associated with Richter’s transformation in patients with chronic lymphocytic leukaemia: protocol for a retrospective population-based cohort study. BMJ Open. 2019;9(3):e023566.
8. Ben-Dali Y, Hleuhel MH, Andersen MA, Brieghel C, Clasen-Linde E, Da Cunha-Bang C, et al. Risk factors associated with Richter’s transformation in patients with chronic lymphocytic leukemia. Blood. 2018;132(Suppl 1):1697.
9. Rossi D, Spina V, Deambrogi C, Rasi S, Laurenti L, Stamatopoulos K, et al. The genetics of Richter syndrome reveals disease heterogeneity and predicts survival after transformation. Blood. 2011;117(12):3391–401.
10. Chigrinova E, Rinaldi A, Kwee I, Rossi D, Rancoita PM, Strefford JC, et al. Two main genetic pathways lead to the transformation of chronic lymphocytic leukemia to Richter syndrome. Blood. 2013;122(15):2673–82.
11. Khan N, Chitalia A, Ozdemirli M, Ray G, Gehan E, Cheson BD. Cell of origin in Richter’s transformation of CLL. Blood. 2012;120(21):4574.
12. Xu L, Song JC, Sun XH, Gao ZF, Lv L, Zhu J. Richter’s syndrome of the central nervous system diagnosed concurrently with chronic lymphocytic leukaemia: a case report and literature review. Medicine (Baltimore). 2018;97(41):e12701.
13. Wang Y, Tschautscher M, Parikh SA, Call TG, Kay NE, Johnston PB, et al. Central nervous system (CNS) involvement of Richter transformation: a single center experience. Blood. 2020;136(Suppl 1):3–4.
14. Ishida F, Nakazawa H, Takezawa Y, Matsuda K, Asano N, Sano K, et al. Richter transformation in the brain from chronic lymphocytic leukemia. J Clin Exp Hematop. 2013;53(2):157–60.
15. Robak T, Góra-Tybor J, Tybor K, Jamroziak K, Robak P, Kordek R, et al. Richter’s syndrome in the brain first manifested as an ischaemic stroke. Leuk Lymphoma. 2004;45(6):1261–7.
16. Villanueva G, Guscott M, Schaiquevich P, Sampor C, Combs R, Tentoni N, et al. A systematic review of high-dose methotrexate for adults with primary central nervous system lymphoma. Cancers (Basel). 2023;15(5):1459.
17. Pronello E, Mo F, Gottardi D, Palmiero R, Bertero L, Lanotte M, et al. Richter’s syndrome of the central nervous system. Can J Neurol Sci. 2021;48(6):889–92.
18. Binet JL, Leporrier M, Dighiero G, Charron D, Vaugier G, Beral HM, et al. A clinical staging system for chronic lymphocytic leukemia. Prognostic significance. Cancer. 1977;40(2):855–64.
19. Rai KR, Sawitsky A, Cronkite EP, Chanana AD, Levy RN, Pasternack BS. Clinical staging of chronic lymphocytic leukemia. Blood. 1975;46(2):219–34.
20. Hans CP, Weisenburger DD, Greiner TC, Gascoyne RD, Delabie J, Ott G, et al. Confirmation of the molecular classification of diffuse large B-cell lymphoma by immunohistochemistry using a tissue microarray. Blood. 2004;103(1):275–82.
21. Lionakis MS, Dunleavy K, Roschewski M, Widemann BC, Butman JA, Schmitz R, et al. Inhibition of B cell receptor signaling by ibrutinib in primary CNS lymphoma. Cancer Cell. 2017;31(6):833–43.e5.
22. Abrey LE, Batchelor TT, Ferreri AJ, Gospodarowicz M, Pulczynski EJ, Zucca E, et al. Report of an international workshop to standardize baseline evaluation and response criteria for primary CNS lymphoma. J Clin Oncol. 2005;23(22):5034–43.
23. Arshad S, Fang X, Ahn KW, Kaur M, Scordo M, Sauter CS, et al. Impact of thiotepa dose-intensity in primary diffuse large B-cell lymphoma of the central nervous system undergoing autologous hematopoietic cell transplant with thiotepa/carmustine conditioning. Bone Marrow Transplant. 2023;58(11):1203–8.
24. Nakamura T, Tateishi K, Niwa T, Matsushita Y, Tamura K, Kinoshita M, et al. Recurrent mutations of CD79B and MYD88 are the hallmark of primary central nervous system lymphomas. Neuropathol Appl Neurobiol. 2016;42(3):279–90.
25. Radke J, Ishaque N, Koll R, Gu Z, Schumann E, Sieverling L, et al. The genomic and transcriptional landscape of primary central nervous system lymphoma. Nat Commun. 2022;13(1):2558.
26. Nishimura N, Takeuchi K, Asaka R, Tuyama N, Inoue N, Kusano Y, et al. MYD88 L265P mutation detected by digital PCR as a prognostic factor in patients with diffuse large B-cell lymphoma in rituximab era. Leuk Res. 2020;97:106426.
27. Phelan JD, Young RM, Webster DE, Roulland S, Wright GW, Kasbekar M, et al. A multiprotein supercomplex controlling oncogenic signalling in lymphoma. Nature. 2018;560(7718):387–91.
28. de Groen RA, Schrader AM, Kersten MJ, Pals ST, Vermaat JS. MYD88 in the driver’s seat of B-cell lymphomagenesis: from molecular mechanisms to clinical implications. Haematologica. 2019;104(12):2337–48.
29. Shuai W, Lin P, Strati P, Patel KP, Routbort MJ, Hu S, et al. Clinicopathological characterization of chronic lymphocytic leukemia with MYD88 mutations: L265P and non-L265P mutations are associated with different features. Blood Cancer J. 2020;10(8):86.
30. Wang W, Zhang Y, Zhao D, Zhang W, Zhou D. Richter’s syndrome in central nervous system with MYD88 L265P and CD79B mutation responded well to ibrutinib containing chemotherapy: a case report and review of the literature. Ann Hematol. 2022;101(8):1869–72.
31. Rossi D, Cerri M, Capello D, Deambrogi C, Rossi FM, Zucchetto A, et al. Biological and clinical risk factors of chronic lymphocytic leukaemia transformation to Richter syndrome. Br J Haematol. 2008;142(2):202–11.
32. Mao Z, Quintanilla-Martinez L, Raffeld M, Richter M, Krugmann J, Burek C, et al. IgVH mutational status and clonality analysis of Richter’s transformation: diffuse large B-cell lymphoma and Hodgkin lymphoma in association with B-cell chronic lymphocytic leukemia (B-CLL) represent two different pathways of disease evolution. Am J Surg Pathol. 2007;31(10):1605–14.
33. Chang H, Cerny J. Molecular characterization of chronic lymphocytic leukemia with two distinct cell populations: evidence for separate clonal origins. Am J Clin Pathol. 2006;126(1):23–8.
34. Zhao Y, Siddiqi I, Wildes TJ, McCracken J, Deak K, Rehder C, et al. Chronic lymphocytic leukemia with two B-cell populations of discordant light chain restrictions in individual patients: parallel development of biclonal B-cell neoplasms or clonal evolution with isotype switch? Am J Clin Pathol. 2023;159(4):337–51.
35. Engel H, Rolink A, Weiss S. B cells are programmed to activate κ and λ for rearrangement at consecutive developmental stages. Eur J Immunol. 1999;29(7):2167–76.
36. Al-Sawaf O, Robrecht S, Bahlo J, Fink A, Cramer P, von Tresckow J, et al. Richter transformation in chronic lymphocytic leukemia (CLL)—a pooled analysis of German CLL Study Group (GCLLSG) frontline treatment trials. Leukemia. 2021;35(1):169–76.
37. Al-Sawaf O, Ligtvoet R, Robrecht S, Stumpf J, Fink AM, Tausch E, et al. Tislelizumab plus zanubrutinib for Richter transformation: the phase 2 RT1 trial. Nat Med. 2024;30(1):240–8.
38. A, Frustaci AM, Condoluci A, Coscia M, Chiarle R, Zinzani PL, et al. Atezolizumab, venetoclax, and obinutuzumab combination in Richter transformation diffuse large B-cell lymphoma (MOLTO): a multicentre, single-arm, phase 2 trial. Lancet Oncol. 2024;25(10):1298–309.
39. Kittai AS, Bond D, Huang Y, Bhat SA, Blyth E, Byrd JC, et al. Anti-CD19 chimeric antigen receptor T-cell therapy for Richter transformation: an international, multicenter, retrospective study. J Clin Oncol. 2024;42(17):2071–9.
40. Smith CJ, Goyal A, Smith BR, Lee D, Jensen A, Alexander J, et al. Lisocabtagene maraleucel for Richter’s transformation—A case series. EJHaem. 2025;6(2):e270011.
41. Siddiqi T, Maloney DG, Kenderian SS, Brander DM, Dorritie K, Soumerai J, et al. Lisocabtagene maraleucel in chronic lymphocytic leukaemia and small lymphocytic lymphoma (TRANSCEND CLL 004): a multicentre, open-label, single-arm, phase 1–2 study. Lancet. 2023;402(10402):641–54.
42. Raizer JJ, Rademaker A, Evens AM, Rice L, Schwartz M, Chandler JP, et al. Pemetrexed in the treatment of relapsed/refractory primary central nervous system lymphoma. Cancer. 2012;118(15):3743–8.
43. Dietrich J, Versmee L, Drappatz J, Eichler AF, Nayak L, Norden A, et al. Pemetrexed in recurrent or progressive central nervous system lymphoma: a phase I multicenter clinical trial. Oncologist. 2020;25(9):747–e1273.
44. Shadman M, Munir T, Robak T, Brown JR, Kahl BS, Ghia P, et al. Zanubrutinib versus bendamustine and rituximab in patients with treatment-naïve chronic lymphocytic leukemia/small lymphocytic lymphoma: median 5-year follow-up of SEQUOIA. J Clin Oncol. 2025;43(7):780–7.
Received: February 17, 2025;
Accepted: March 18, 2025;
Published: March 20, 2025 .
To cite this article : Steger J, Purow J, Al Sagheer T, Perez Perez A, Odia Y, Chen Z, et al. Efficacy of the Novel Regimen Pemetrexed-Zanubrutinib in a Patient with Chronic Lymphocytic Leukemia and Central Nervous System Diffuse Large B Cell Lymphoma Presenting as Richter’s Transformation. British Journal of Cancer Research. 2025; 8(2): 802- 808. doi: 10.31488/bjcr.207.
© The Author(s) 2025. This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/).
