Research article / Open Access
DOI: 10.31488/bjcr.209
New Approach for Recruiting Medically Underserved Populations to Community-Based Participatory Research
Jenny Ostien, Lauren McCauley-Hixenbaugh, Amy Allen, Stephenie Kennedy-Rea
WVU Cancer Institute, Department of Cancer Prevention and Control, Mobile Cancer Screening Program, PO Box 9350, Morgantown, WV 26506 WV, United States of America
*Corresponding author:Jenny Ostien, MS, WVU Cancer Institute, Department of Cancer Prevention and Control, Mobile Cancer Screening Program, PO Box 9350, Morgantown, WV 26506 WV, United States of America, phone: 304-293-0482, fax: 304-293-6333, jostien@hsc.wvu.edu, 0000-0001-6001-5077.
Abstract
Background: Disparities exist in all parts of the healthcare continuum. Minority and underserved populations including rural, women, aging, non-white, and LGBTQ+ individuals are underrepresented in clinical research. Mobile healthcare units improve access for populations facing barriers. Our goal was to determine if mobile healthcare could also be used as an effective method of improving access to clinical research. Problem: Clinical research is traditionally conducted in a medical facility in a highly populated area leaving people with access barriers unable to participate. Clinical research also leaves the genetic and social makeup of the medically underserved from consideration when developing new interventions. Patients are often misinformed about how they function, the purpose they serve, and their benefit to community members. Methods: Leveraging both our mobile breast and lung cancer screening units, the WVUCI amplified healthcare options in rural, Appalachian communities in West Virginia with exposure to research opportunities. These community-based participatory research projects provided patients access to not only clinical research but also education on different types of research. The units conducted five small-scale research projects. Projects were evaluated retroactively to determine the effectiveness of this new approach. Results/Conclusions: Through the five projects, 751 patients were enrolled and over 1,000 were provided education and exposure to clinical research. By leveraging the existing trust-filled relationship patients have with the mobile unit’s staff increased patient knowledge, understanding, and potential acceptance of future research opportunities. This new model showcases a way to integrate clinical research into existing patient care workflows in a mobile healthcare setting.
Key Words: mobile health screening, clinical trials, innovation, medically underserved populations, rural population
Introduction
Clinical Trials and Barriers to Participation
Clinical trials are considered the gold standard in solving public health problems yet not all populations are appropriately represented [1]. Disparities in the recruitment of rural, women, aging, minorities, and LGBTQ+ individuals to clinical trials persist [2]. This lack of representation is alarming because it omits specific health conditions, genetic markers, and environmental exposures from data sets used to develop screening and treatment recommendations and health policies. While these underrepresented populations are subjected to recommendations developed for a general population that may not benefit them, it also costs payors an exorbitant amount of money that could be avoided with better representation when developing recommendations [2]. Researchers assume the lack of participation by racial minorities and rural residents is because these populations are not willing to participate but evidence suggests that if they are asked, they are willing to participate. [2, 3]. Our research aimed to discover what factors hindered these underrepresented populations from participating in research.
Common barriers to clinical research participation include transportation, access to opportunities, associated costs, time commitment, conflict with familial responsibilities, work responsibilities, insufficient knowledge about research (including being unfamiliar with anything other than treatment trials [4]), and location of research centers. Several misconceptions were also cited as reasons for not participating in clinical research including the fear of being given a placebo, losing control over decisions regarding their health, fear they would be part of a randomized group and not receive quality care, and thinking that participating in research was a “last-ditch” effort to help them [5].
Barriers for Medically Underserved Populations in Clinical Research – West Virginia, Rural Appalachia Cohort Experience
Residents of Appalachia are typically considered medically underserved, and cancer mortality rates are higher for Appalachian residents in comparison to the rest of the United States (US). Financial and geographic barriers to care, education level, high rates of unemployment, low per capita income, lack of public transportation, cost of travel and personal transportation, travel time to care, and the low number of primary care and specialty care providers [6, 7] are barriers that exist for patients in Appalachia even before being approached about participating in clinical trials.
West Virginia (WV) is the only state that lies entirely within Appalachia and is considered the third most rural state in the nation, with 61.8% of the state’s counties designated as rural [8] and over half (51.8%) of residents living in rural areas where lack of reliable transportation and lengthy driving times contribute to delays in health screening and follow-up care [9]. WV ranks first in the US for colorectal and laryngeal cancer deaths, second in the US for lung and oral cancer deaths, and third in the US for overall cancer deaths [10]. While the combined incidence rates for cancer are higher in urban areas, mortality rates are higher in rural areas. Recently, death rates from cancer decreased in both rural and non-rural areas, but the rate of decline was slower in rural areas, increasing the differences in mortality [11].
West Virginia University Mobile Cancer Screening Program
Mobile healthcare units have improved access for populations facing a variety of barriers to healthcare including transportation, access to services, and funding for the underinsured or uninsured. Access helps improve health equity and is an opportunity to increase access to and participation in clinical research. Using two mobile cancer screening units, the West Virginia University Cancer Institute (WVUCI) Mobile Cancer Screening Program (Program) enhances healthcare options for rural communities by offering patients breast and lung cancer screening and access to clinical research opportunities. The WVUCI provides world-class cancer screening to communities in need by growing its network across the region and state. This approach provides patients with decreased travel time, a reduction in the amount of time off work, increased access to cancer screening and early detection, and financial support through grants and donations to fund screening for eligible residents who are underinsured or uninsured. Working with local clinics, businesses, and healthcare providers, Bonnie’s Bus Mobile Mammography Unit (Bonnie’s Bus) and the Mobile Lung Cancer Screening Unit (LUCAS), travel across WV offering cancer screening services where it is convenient for patients--in their communities. Both units are led by WVUCI Department of Cancer Prevention and Control (CPC) staff and are operated by WVU Hospitals.
WVUCI Department of Cancer Prevention and Control Work with Communities
Cultivating relationships with communities is the cornerstone of all CPC endeavors. Instead of taking fully developed projects into a community, CPC engages its partners early in the process so they can aid in their development. This allows the partners to acknowledge local barriers to healthcare, address cultural intricacies, and use known methods of reaching their patient population. CPC also develops health literate, culturally appropriate education and outreach initiatives; conducts community-based participatory research; translates cancer-related science and research for WV communities; and connects WV communities to WVU and the WVUCI. The existing Program infrastructure combined with CPC and its history of community engagement led the team to seek opportunities to expand its services to patients during their annual cancer screening appointments. One area that was identified was clinical research. Clinical research is traditionally conducted in a medical or university setting but often does not take into consideration the social determinants of health and the conditions in the environment where people are born, live, work, play, and worship, which affect participation and health outcomes [12]. This approach addresses previous research that revealed, “…increasing rural patients’ access to care requires expanding services and decreasing travel distance, mitigating financial burdens when insurance coverage is limited, opening avenues to clinical trial participation, and creating partnerships between providers and community leaders to address local gaps in care [6].”
The Program hypothesized that it could use mobile units to overcome several patient barriers to research. The Program expanded existing partnerships and fostered new ones to conduct five small-scale studies to not only provide opportunities to patients but to test the feasibility and acceptability of this approach.
Partner descriptions
Partnerships are pivotal to engaging communities in the unique approach of using the Program to take clinical research to rural communities that have had limited exposure to research. Even though CPC has approximately 700 partnerships, 5 specific projects with the American Cancer Society, Breathe BioMedical™, Epigenomics, the Mobile Health Innovation Collaborative, and Mountains of Hope (West Virginia’s Centers for Disease Control and Prevention funded Comprehensive Cancer Coalition) are highlighted here.
- The American Cancer Society (ACS) works to improve the lives of people living with cancer and their families. They do this through advocacy, research, and patient support to ensure everyone has access to cancer resources. ACS introduced new partners, personnel, and supplies.
- Breathe BioMedical™ seeks ways to make early disease screening of cancer and other diseases easier through breath analysis as the new standard for routine screening. The technology works by identifying the biomarkers of disease in breath samples. Breathe BioMedical™ provided staff training, equipment, supplies, and screening test analysis.
- Epigenomics introduced the first FDA-approved blood test (Epi proColon®) for colorectal cancer (CRC) screening for people who are not up to date on CRC screening or who are unwilling or unable to complete guideline-recommended screening. Epigenomics provided staff training, supplies, and specimen analysis.
- The Mobile Health Innovation Collaborative (MHIC) was developed by the Mobile Healthcare Association and Harvard Medical School’s Mobile Health Map to help organizations operating mobile programs measure, evaluate, and communicate the impact mobile health services have on patients and the broader community. MHIC provided funding, evaluation education, and mentoring.
- Mountains of Hope (MOH) is West Virginia’s state cancer coalition and is dedicated to cancer prevention, early detection, and improving the quality of life of West Virginians affected by cancer. MOH includes more than 150 healthcare professionals, cancer survivors, and community advocates who help provide resources, support groups, financial assistance, and transportation for West Virginians affected by cancer. MOH provided introduction to new partners, personnel, resources, and supplies.
Methods
Addressing health inequities and providing cancer screening are two pillars of the Program. Substantial planning led to the implementation of community-based participatory research on the mobile units to expose patients to research opportunities. Staff worked with their community partners to gauge interest, identify recruitment strategies, develop appropriate materials, and review the roles and responsibilities of each team member. Community partners were included early in the development of each project to account for local needs. This facilitated tailored recruitment tactics as the study team was aware that effective strategies in one community could potentially be less effective in other communities. Allowing flexibility for recruitment approaches, messaging, and outreach to each partner allowed for a personalized approach.
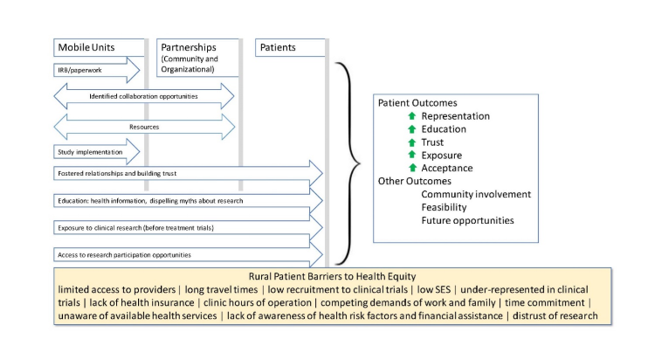
Figure 1:Model workflow to use mobile healthcare units to take research into communities.
The model (Figure 1) of utilizing mobile cancer screening units to conduct population-based research began with identifying potential community resources and procuring both human and financial resources. Assembling a unified team and delineating roles was essential to these community partnerships and the development of Institutional Review Board (IRB) protocols. Program staff were encouraged to simultaneously foster relationships and build trust with all parties involved throughout study implementation. These relationships provided educational opportunities and access to clinical trials which in turn helped to dispel myths about clinical trials.
Results
Each of the research projects had its own measure of success based on the specific aims of the respective project. However, the success of conducting research in Appalachia cannot be measured by quantitative data alone. Qualitative comments from participants, community members, and staff provided valuable insight regarding participation in clinical research. Individually, the results are interesting, but together they tell a richer story.
The Program patients became excited to contribute and readily accepted the invitation to participate in community-based participatory research conducted on the mobile units. (Figure 2). Appalachians are known for their sense of place and community and those values were evident as many of the patients in these projects said, [they] hope that [their] contribution helps one of [their] neighbors. Participants learned the value of partaking in research to find less invasive ways of screening and in creating educational and awareness opportunities for fellow Appalachians. They discovered that their experience contributes to opportunities beyond Appalachia and helps on a national scale. A secondary outcome observed through these projects is that many participants expressed increased trust in the healthcare system because of their inclusion in these opportunities.
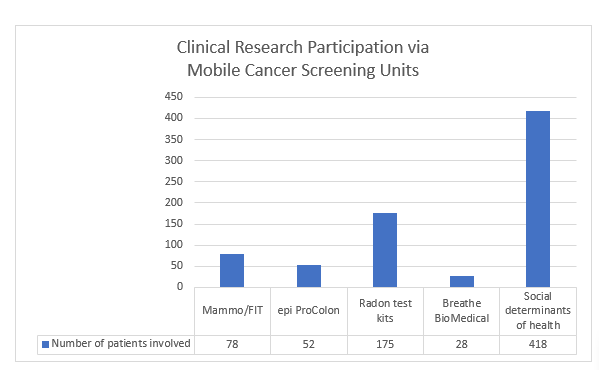
Figure 2:Community participation from mobile healthcare research projects.
The trust and access created by utilizing our existing resources demonstrated the merit of this type of implementation science to each partner involved. A total of 751 people participated in clinical research through the five efforts focusing on taking research to underserved populations. Community providers have indicated that they have seen growth in their patient populations as well as an increase in completing screening follow-up recommendations. Mobile unit staff have seen patient interest in research increase, and several have asked if there are new opportunities to participate when they come for their cancer screening appointment. Our principal outcomes demonstrate there are many opportunities for growth and future collaboration.
As mentioned, the qualitative information gathered was insightful and added meaningful context to the overall story. Patient interest in clinical research was higher than initially expected and when talking with patients they stated that they wanted to participate for several reasons:
• Desire to help the mobile unit out (related to a long-standing relationship and trust in our unit)
• Excitement to be included in something new that they have not been exposed to previously
• Sense of purpose and family responsibility; even though they may not benefit directly from participating, knowing that it may help their children, grandchildren, or neighbors, makes participation worthwhile
While most patients come from rural, medically underserved areas with poor cancer health outcomes, negative behavioral contributions, and disparities that compound these outcomes, there is more to their story. Appalachian values of independence, family, community, hard work, and resilience are strengths that help them overcome and adapt. These values and the strong connection to the community likely contributed to their participation in a clinical trial or research study.
Discussion
The Program’s mission combines care close to home with clinical research and the desire to provide patients with as many health-related opportunities as possible to reduce health inequities. Social determinants of health in WV, specifically rurality, education level, and a high number of areas designated as medically underserved, give this type of innovative approach a strong foothold in the state. Mobile healthcare has become a trusted partner in hundreds of communities across WV and introducing clinical research and clinical trials to these populations was an organic fit. While literature reviews indicate few examples of mobile healthcare being utilized for research opportunities, it did indicate the importance of representation. Ethnic diversity in recruitment is a vital prerequisite to eliminating health disparities in cancer treatment, control, and prevention programs [13].
Historically, patients only receive information about cancer clinical trials after receiving a diagnosis such as cancer at a time when they are overwhelmed with information and emotion. Some misconceptions are still prevalent about clinical trials including that by participating they will not receive the same level of care as if they were not on a trial, they will receive placebo medications, they will be a “Guinea Pig”, or that their disease is so severe the clinical trial is a “last-ditch” effort [14, 5]. Most people do not realize that prevention and early detection trials exist [4]. Through interactions with mobile patients since 2009, it has been noted repeatedly that they are not accessing the services (offered on the mobile units) at traditional facilities. Based on the approach defined in the methods section, an argument can be made that using mobile healthcare units to take research into communities, will:
• Increase exposure and knowledge of clinical research before a cancer diagnosis,
• Increase exposure and knowledge of prevention and early detection research,
• Expand geographical reach,
• Dispel myths about clinical trials,
• Increase the number of participants from special populations participating in research,
• Speed up accruals,
• Advance precision medicine because of the diversity of data collected and faster accrual period,
• Navigate patients toward a more stable medical home, and
• Enhance engagement within communities and community partners.
It is also important to take into consideration the stress additional projects on the mobile units may create. Specifically, overworking employees, decreasing the amount of time for the original service, decreasing the quality of service to patients, and uncertainty of patient responsiveness to the research. However, with advanced training and alternative patient flow protocols, these barriers are easily addressed. The Program ensures all staff members are CITI trained and participate in thorough protocol training, as well as engaging them in the development of alternative workflows for study participants accessing screening and participating in research. Staff are part of the process and help ensure the integration of the research into their existing procedures to make the transition as minimally disruptive as possible. Talking about how to incorporate the research into the workflow of the mobile staff starts early and is routinely readdressed to ensure maximum efficiency and patient care. This process is reviewed and adapted as data is gathered in the field. Staff are excited about being part of the research team and participating in something different from their typical day-to-day responsibilities and concern about the increased work burden have not been issues.
While the use of mobile healthcare units improves access for rural populations to clinical research, limitations still exist and need to be acknowledged. Staff working on the five projects noted that while the mobile units decreased the barrier to research locations, they cannot completely solve transportation barriers using the current model. The use of the mobile cancer screening units also only reaches individuals who are actively seeking healthcare, those eligible for the cancer screenings being offered by the units, and those with the ability to complete the informed consent independently. The complexity, space requirements, and trials that require multiple visits may not fit into the workflow of existing mobile unit services. Specialty practitioners needed for a study (MD, APP, NP, phlebotomist, etc.) increase the number of people on a unit and the cost associated with the project. Equipment needed for the study (refrigeration, freezing, hazardous materials, centrifuge, etc.) may not be feasible in a mobile unit. However, many of the limitations can have creative solutions and workarounds to engage populations with limited access to healthcare and those inadequately represented in clinical research.
Conclusion
Using mobile healthcare units is an effective way to conduct clinical research while providing high-quality services to patients. It is both a feasible and acceptable method of addressing transportation issues, access to opportunities, and increasing underrepresented demographic patient access to cutting-edge research and clinical trials. Mobile services with clinical research on board can serve a myriad of underserved patient populations such as rural, women, aging, minority, and LGBTQ+ or those that may be specific to clinical trials research. This approach can foster not only underserved and minority recruitment to clinical trials but also act as a gateway to healthcare options and medical homes. When conducted appropriately it is not a burden to staff and does not negatively impact the high quality of health services patients on the mobile unit are accustomed to receiving. The model can be replicated on any mobile unit already in practice without diluting its primary mission. This partnership creates benefits well beyond its positive impact on patients and healthcare systems. Communities, community members, public health officials, and providers could see the residual benefits of having local representation in clinical research. Finally, it introduces the language of clinical trials to patients before a stressful health condition occurs with the hope it will enable the patient to be more receptive to conversations about interventional clinical trials. Broadening the depth and breadth of existing research is beneficial to the community, healthcare system, state, and potentially the global research community. The expansion of clinical research services offered on mobile units is a tremendous opportunity for growth. The studies conducted were limited pilots but could be expanded to provide larger trials with a greater impact on the healthcare industry. Expanding the service network through mobile clinical trial research can allow for greater dissemination of vital health information to the community and expand current patient materials and communications. Overall, using mobile units for research is a symbiotic relationship between patients and the healthcare entity.
Acknowledgments
Kyle Chapman, MD, Michelle Chappell, Hannah Hazard-Jenkins, MD, FACS, Harvard Health Map, Mobile Healthcare Association, Mobile Health Innovation Collaborative, WVU Cancer Institute Department of Cancer Prevention and Control Staff.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
ACS: American Cancer Society; CPC: Cancer Prevention and Control; CRC: colorectal cancer; IRB: Institutional Review Board; LGBTQ+: lesbian, gay, bisexual, transgender, queer/questioning; LUCAS: mobile lung cancer screening (unit); MHIC: Mobile Healthcare Innovation Collaborative; MOH: Mountains of Hope; US: United States; WV: West Virginia; WVU: West Virginia University; WVUCI: West Virginia University Cancer Institute;
References
1. Wallington SF, Dash C, Sheppard VB. Enrolling minority and underserved populations in cancer clinical research. Am J Prev Med. 2016;50(1):111–7.
2. National Academies of Sciences, Engineering, and Medicine. Improving representation in clinical trials and research. Washington (DC): National Academies Press; 2022.
3. Kennedy-Rea S, Mason J, Hereford C. Engaging community perceptions of research in rural West Virginia. Collaborations. 2021;4(1):Article 6.
4. Hall MJ, Egleston B, Miller SM. Barriers to participation in cancer prevention clinical trials. Acta Oncol. 2010;49(6):757–66.
5. National Institutes of Health, Office of Research on Women’s Health. Review of the literature: primary barriers and facilitators to participation in clinical research [Internet]. Bethesda (MD): NIH; [cited 2025 Aug 2]. Available from: https://orwh.od.nih.gov
6. Levit LA, Byatt L, Lyss AP. Closing the rural cancer care gap: three institutional approaches. JCO Oncol Pract. 2020;16(7):422–30.
7. Lengerich EJ, Wyatt SW, Rubio A. The Appalachia Cancer Network: cancer control research among a rural, medically underserved population. J Rural Health. 2004;20(2):181–7.
8. West Virginia Center on Budget & Policy. State of rural West Virginia [Internet]. Charleston (WV): WVCBP; 2018 Oct [cited 2025 Aug 2]. Available from: https://wvpolicy.org
9. Rural Health Information Hub. State rural health plans.: RHIhub; [cited 2025 Aug 2]. Available from: https://www.ruralhealthinfo.org
10. Beskow LM. Lessons from HeLa cells: the ethics and policy of biospecimens. Annu Rev Genomics Hum Genet. 2016;17:395–417.
11. Henley SJ, Anderson RN, Thomas CC. Invasive cancer incidence, 2004–2013, and deaths, 2006–2015, in nonmetropolitan and metropolitan counties—United States. MMWR Surveill Summ. 2017;66(14):1–13.
12. US Department of Health and Human Services. Social determinants of health—Healthy People 2030 [Internet]. Washington (DC): HHS; . Available from: https://health.gov/healthypeople/objectives-and-data/social-determinants-health
13. Bruner DW, Jones M, Buchanan D. Reducing cancer disparities for minorities: a multidisciplinary research agenda to improve patient access to health systems, clinical trials, and effective cancer therapy. J Clin Oncol. 2006;24(14):2209–15.
14. Johns Hopkins Medicine. Clinical trials: separating fact from fiction.
Received: March 06, 2025;
Accepted: March 27, 2025;
Published: April 02, 2025 .
To cite this article : Ostien J, McCauley-Hixenbaugh L, Allen A, Kennedy-Rea S. New Approach for Recruiting Medically Underserved Populations to Community-Based Participatory Research. British Journal of Cancer Research. 2025; 8(2): 816- 821. doi: 10.31488/bjcr.209.
© The Author(s) 2025. This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/).
