Research article / Open Access
DOI: 10.31488/bjcr.208
Socioeconomic Differences in Colorectal Cancer Mortality: A Population-Based Analysis Using the Office for National Statistics Colorectal Cancer Mortality Data for England, 2011-2020
Emma Taylor-Gallardo*, Eva Morris, Stephanie Ross, Nick Hall,Anjum Memon, Nazrul Islam
1. Department: Big Data Institute University of Oxford, Old Road Campus, Oxford, OX3 7LF
*Corresponding author:Emma Taylor-Gallardo. Department: Big Data Institute University of Oxford, Old Road Campus, Oxford, OX3 7LF, email: emma.taylor-gallardo1@nhs.net.
Abstract
Background: Socioeconomic inequalities in colorectal cancer (CRC) mortality have previously been reported in England, linked to modifiable risk factors, engagement with screening programmes and unequal access to treatment services. Methods: A population-based retrospective analysis of the Office for National Statistics data for CRC deaths in England for 2011-2020 was conducted to examine the association between area-based socioeconomic deprivation and CRC mortality, by age and sex. Eligible records with CRC (ICD-10 C18-20) as an underlying or contributing cause of death were assigned to one of five deprivation groups using the English Index of Multiple Deprivation. Crude and age-specific rates were calculated. Age-standardised mortality rates (ASMR) were calculated using direct standardisation using the 2013 European Standard Population. Rates were calculated by year of death, age, sex and level of deprivation and reported per 100,000 people. Poisson regression models by year of death, age, sex and level of deprivation were constructed to examine the difference in the incidence rate ratio of mortality between the most and least deprived groups. Results: Amongst the 151,050 records eligible for analysis, people in the most deprived areas experienced a 21-28% higher CRC mortality rate than those in the least deprived. The difference in inequalities was greater in males (29-46%) than females (9-20%). The ASMR of CRC was higher in males than to females. Conclusions: We observed differences in CRC mortality rates in England by deprivation and sex. The greatest inequalities were seen in males from the most deprived areas. Further research into underlying causes is required to improve the understanding of the association between socioeconomic deprivation and CRC mortality.
Keywords: colorectal cancer, mortality, socioeconomic differences
Introduction
According to GLOBOCAN 2020, colorectal cancer (CRC)(ICD-10 code C18-20) is the third most commonly diagnosed cancer and the second most common cause of cancer mortality worldwide, with over 1.9 million incident cases and around 930,000 deaths [1].
The global distribution of CRC burden varies widely, with known gradients in incidence and mortality across human development levels; more than two-thirds of cases and around 60% of deaths occur in high-income countries [2]. Understanding the temporal trends and demographic distribution of CRC is important as the global incidence is expected to increase over the coming decade to more than 2.2. million new cases and 1.1 million deaths in 2030 [2].
Trends in the UK are similar to other western countries. In the UK CRC was the 4th most common cancer and 2nd most common cause of cancer death [3], with around 43,000 new diagnoses and 16,800 deaths in 2018. CRC incidence rates in the UK have remained relatively stable over the last two decades, with a 6% decrease in the incidence rate (European age-standardised).
Survival has more than doubled in the last 40 years, with a 53% 10-year survival. Five-year net survival for CRC ranges from 92% for those diagnosed at stage 1, and 10% for those diagnosed at stage 4[3]. Mortality rates have decreased by around 11% in the UK in the last decade and decreased by 45% since the early 1970s. This is in part due to stabilisation of incidence rates and improvements in survival through early detection by screening programmes and improvements in treatment options [4].
In high income countries such as England, there is a known difference in the incidence, survival and mortality from CRC by sex and deprivation. Incidence and mortality rates are higher in males than females, and in people living in more deprived areas compared to less deprived areas [5,6].
CRC mortality is known to be reduced through participation in screening programmes through early detection [7]. The English Bowel Cancer Screening programme, first introduced in 2006, now involves the use of the Faecal Immunochemical Test (FIT). It is offered every two years to men and women aged 60 to 74 [8]. Removal of precursor and neoplastic CRC lesions have been shown to reduce future CRC incidence. Uptake of screening is estimated to vary between 35% in the most deprived and 61% in the least deprived regions of the UK [9].
The aetiology of CRC is fairly well understood - 54% of CRC are thought to be preventable [3]. Non-modifiable risk factors include age, family history and genetic factors including hereditary non-polyposis colorectal cancer (HNPCC) and familial polyposis10. Modifiable risk factors include obesity, sedentary lifestyle, processed and red meat consumption, alcohol and tobacco use [4].
Socioeconomic deprivation is a multi-faceted concept that refers to the complex factors that influence an individual’s experience in society. Socioeconomic deprivation in England can be measured using the English Index of Multiple Deprivation (IMD) [11]. It is an index measure comprised of seven domains that provides an overall relative measure of deprivation for each Lower layer Super Output Area (LSOA), which accounts for a small area with a population of around 1,500 people. Importantly, the IMD score is an area-based score and does not provide information on whether everyone within an LSOA experiences the same level of deprivation.
Previous research has investigated socioeconomic inequalities in CRC screening uptake [12] and mortality rates in England [5-13]. The most deprived areas of England experience higher incidence and mortality rates of common cancers such as bowel, breast and prostate cancer compared to those in less deprived areas [5]. The reasons for these differences are thought to come under the domains defined by IMD as well as differences in health related behaviours such as smoking, alcohol consumption, sedentary lifestyle, nutrition, access to and interaction with healthcare services [14].
This study aims to investigate the association between socioeconomic deprivation and CRC mortality in England, using CRC mortality data for the years 2011-2020. A population-based analysis of the Office for National Statistics (ONS) mortality data will be conducted with the aim of improving understanding of the association between socioeconomic deprivation and CRC mortality
Methods
To examine the association between IMD and CRC mortality, a population-based retrospective epidemiological study, using ONS mortality data, was conducted using records of CRC (ICD-10 C18-20) as the underlying or contributing cause of death between 2011 and 2020 in England. For the calculation of mortality rates, English population data by age, sex and calendar year (i.e., the denominator data) were obtained from ONS census estimates. The ONS dataset included information on CRC deaths in England, age at death, sex and socioeconomic deprivation.
IMD is the official measure of relative deprivation for small areas in England, based on the 2011 census [15]. The IMD ranks every small area of around 1,500 people from 1 (most deprived) to 32,844 (least deprived) using seven domains of deprivation (see supplementary). IMD rank was collapsed into IMD quintiles for analysis.
Age-specific and age-standardised mortality rates (ASMR) for CRC were calculated for England (population 56.5M) by year and sex, overall and by IMD quintiles. Rates were reported per 100,000 people. Age standardisation was done using the European Standard Population 2013 Edition [16].
In order to determine the difference in mortality rates between each IMD quintile, age-adjusted incidence (mortality) rate ratios were calculated using a Poisson count regression model for each year, using the least deprived quintile (IMD 5) as the reference category. Age was adjusted for as confounder and the model was stratified by sex. Logarithm of the population was used as the offset term.
There was evidence of over-dispersion which violates the assumptions of the Poisson model. Therefore, robust variance estimators were used to calculate heteroscedasticity-robust standard errors, accounting for Poisson model misspecification. However, we also checked the robustness of the results from the Poisson model with robust variance in the sensitivity analysis using a negative binomial model. Since level of deprivation by IMD quintile is the exposure of interest, we conducted a linear test for trend on the association between CRC mortality and decreasing deprivation by sex and for each year.
All data management and analyses were conducted using STATA v.1. A separate ethics approval was not required because only anonymous data without personal identifiers was used in this analysis.
Results
In the 10-year period 2011-2020, there were 151,302 patient records obtained from the ONS database where a death due to CRC was recorded. Of these, 252 were excluded from the analysis as these deaths occurred in Scotland and Wales. The final analysis included 151,050 CRC deaths in England. There were no missing data.
Table 1 shows higher rates of CRC mortality in males than females across all years 2011 to 2020. Although there was an increase in the number of cases of CRC deaths in England from 2011 to 2020, the crude rates for CRC mortality were fairly stable.
Table 1. Crude and age standardised colorectal cancer mortality rates by sex in England, 2011-2020
| Year | Deaths | Crude* | ASR (95% CI)** |
|---|---|---|---|
| Males | |||
| 2011 | 7,861 | 30.1 | 39.8 (38.9-40.7) |
| 2012 | 8,245 | 31.3 | 40.8 (39.9-41.7) |
| 2013 | 8,115 | 30.6 | 39.3 (38.4-40.1) |
| 2014 | 8,037 | 30.0 | 38.1 (37.2-38.9) |
| 2015 | 8,142 | 30.1 | 37.6 (36.8-38.4) |
| 2016 | 8,303 | 30.4 | 37.3 (36.5-38.2) |
| 2017 | 8,503 | 30.9 | 37.2 (36.4-38.0) |
| 2018 | 8,488 | 30.7 | 36.3 (35.5-37.1) |
| 2019 | 8,665 | 31.1 | 36.2 (35.4-36.9) |
| 2020 | 9,006 | 32.2 | 36.9 (36.2-37.7) |
| Females | |||
| 2011 | 6,476 | 24.0 | 24.1 (23.5-24.7) |
| 2012 | 6,729 | 24.8 | 24.7 (24.1-25.3) |
| 2013 | 6,651 | 24.3 | 24.1 (23.5-24.7) |
| 2014 | 6,614 | 24.0 | 23.5 (23.0-24.1) |
| 2015 | 6,702 | 24.1 | 23.5 (23.0-24.1) |
| 2016 | 6,821 | 24.4 | 23.7 (23.1-24.3) |
| 2017 | 6,692 | 23.8 | 22.8 (22.2-23.3) |
| 2018 | 6,849 | 24.2 | 22.9 (22.4-23.5) |
| 2019 | 6,994 | 24.6 | 23.0 (22.5-23.5) |
| 2020 | 7,157 | 25.1 | 23.4 (22.8-23.9) |
| Persons | |||
| 2011 | 14,337 | 27.0 | 30.8 (30.3-31.3) |
| 2012 | 14,974 | 28.0 | 31.6 (31.1-32.1) |
| 2013 | 14,766 | 27.4 | 30.7 (30.2-31.2) |
| 2014 | 14,651 | 27.0 | 29.8 (29.3-30.3) |
| 2015 | 14,844 | 27.1 | 29.6 (29.1-30.1) |
| 2016 | 15,124 | 27.4 | 29.6 (29.2-30.1) |
| 2017 | 15,195 | 27.3 | 29.1 (28.6-29.5) |
| 2018 | 15,337 | 27.4 | 28.8 (28.3-29.3) |
| 2019 | 15,659 | 27.8 | 28.8 (28.3-29.2) |
| 2020 | 16,163 | 28.6 | 29.4 (28.9-29.8) |
* Crude colorectal cancer mortality rate per 100,000
** Age standardised colorectal cancer mortality rate per 100,000
There was a modest decline in the ASMR of CRC mortality in males, females, and persons. In males, there was a modest decline of 7.3% in the ASMR of CRC mortality from 39.8 (95% CI 38.9-40.7) in 2011 to 36.9 (95% CI 36.2-37.7) in 2020. In females, there was also a small decrease of 2.9% in the ASMR of CRC mortality from 24.1 (95% CI 23.5-24.7) in 2011 to 23.4 (95% CI 22.8-23.9) in 2020.
Figure 1 shows the age-specific CRC mortality rates by year of death and sex in England for 2011-2020. Older age groups have a higher CRC mortality rate in males, females, and persons. Males had a pronouncedly higher age-specific CRC mortality rate than females across older age categories (60-69, 70-79, 80-89 and 90+). This difference was particularly prominent in females in the age category 90+, who had a similar age-specific CRC mortality rate to males in the age category 80-89.
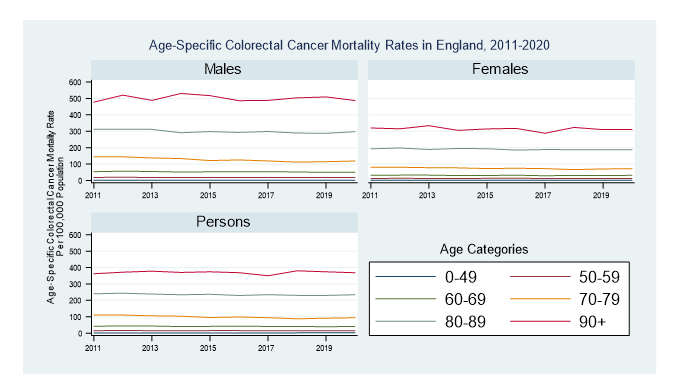
Figure 1:Age-specific colorectal cancer mortality rates by year of death and sex in England, 2011-2020
Supplementary table 1 shows that males had higher age-specific CRC mortality rates than females at all ages except in the age 0-49 group, where the rates were similar.
Overall, age-specific CRC mortality rates increased with the level of deprivation. The exceptions to this are seen in the youngest and oldest age categories (0-49 and 90+), where mortality rates were similar across deprivation quintiles. The greatest differences in age-specific mortality rates by deprivation were seen in the age categories 70-79 and 80-89.
In 2020, males aged 80-89 in the most deprived areas had an increased age-specific CRC mortality rate of 22.4% compared to males in the least deprived areas (345.26 in the most deprived areas vs 268.08 in the least deprived areas). In the same year, females ages 80-89 in the most deprived areas had an increased age-specific mortality rate of 10.9% compared to females in the least deprived areas (195.34 in the most deprived areas vs 174.12 in the least deprived areas).
In those aged 90+ there was a reversal in the direction of association between deprivation and mortality. In 2011 those ages 90+ in the most deprived areas had the highest mortality rate, however by 2020 this was reversed and those in the less deprived areas had a higher mortality rate.
Figure 2 shows the ASMR mortality rates by year of death, sex and IMD in England, 2011 to 2020. Rates were higher for males than females across all areas of deprivation. The ASMR in males in the most deprived areas was almost double that of women in the most deprived areas. There was a greater inequality in ASMR of CRC mortality by level of deprivation amongst men than in women.
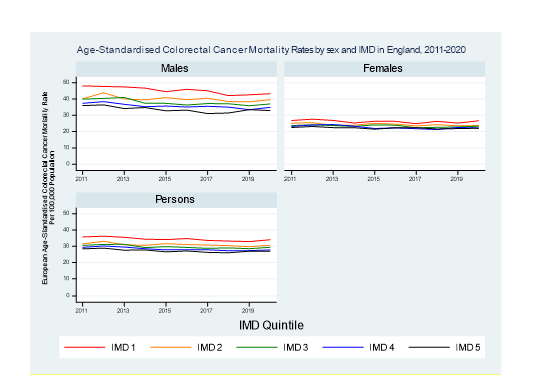
Figure 2:Age-standardised colorectal cancer mortality rates by year of death, sex and IMD in England, 2011-2020
Supplementary table 2 shows that those in more deprived areas have a lower crude CRC mortality rate than those in the less deprived areas. Once standardising for age, the direction of this association is reversed and those in the most deprived areas have higher ASMR CRC mortality. ASMR are higher in males than in females at all levels of deprivation, and the difference in ASMR of mortality by level of deprivation are greater in males than in females. In 2020, males in the most deprived areas had a 24.0% increased ASMR of mortality compared to males in the least deprived areas (43.3 in the most deprived areas vs 32.9 in the least deprived areas). In 2020, females in the most deprived areas had an increased ASMR of 17.2% (26.7 in the most deprived areas vs 22.1 in the least deprived areas areas).
Table 2 shows the incidence rate ratio of mortality (IRR) for those in the most deprived areas (IMD 1) compared to those in the least deprived areas (IMD 5) as baseline. Males in IMD 1 had a higher IRR than those in IMD 5. During the 10-year study period, males in the most deprived areas had an estimated 29-46% increased risk of mortality from CRC compared to males in the least deprived areas. Whereas in females in the most deprived areas, this risk varied from 9-20% compared to females in the least deprived areas. These data also suggest that males in deprived areas are relatively more affected by deprivation on CRC mortality than females. In 2020, males in the most deprived areas had a 35% increased mortality rate ratio than males in the least deprived areas (IRR 1.35 95% CI 1.23-1.47). In 2020, females had a 20% increased IRR than females in the least deprived areas (IRR 1.20 95% CI 1.07-1.34).
Table 2. Shows the IRR and 95% CI for those in IMD 1 (most deprived) compared to those in IMD 5 (least deprived)
| IRR (95% CI) | |||
|---|---|---|---|
| Males | Females | Persons | |
| 2011 | 1.35 (1.22-1.49) | 1.17 (1.07-1.28) | 1.25 (1.13-1.38) |
| 2012 | 1.34 (1.23-1.47) | 1.17 (1.08-1.28) | 1.23 (1.14-1.37) |
| 2013 | 1.39 (1.27-1.51) | 1.17 (1.03-1.33) | 1.27 (1.14-1.41) |
| 2014 | 1.35 (1.25-1.47) | 1.12 (1.05-1.19) | 1.23 (1.15-1.33) |
| 2015 | 1.37 (1.28-1.47) | 1.20 (1.04-1.37) | 1.28 (1.16-1.41) |
| 2016 | 1.40 (1.28-1.53) | 1.16 (1.05-1.27) | 1.28 (1.16-1.40) |
| 2017 | 1.46 (1.35-1.58) | 1.09 (0.98-1.22) | 1.27 (1.16-1.40) |
| 2018 | 1.35 (1.20-1.52) | 1.20 (1.07-1.35) | 1.27 (1.13-1.43) |
| 2019 | 1.29 (1.17-1.42) | 1.14 (1.05-1.24) | 1.21 (1.12-1.31) |
| 2020 | 1.35 (1.23-1.47) | 1.20 (1.07-1.34) | 1.27 (1.15-1.40) |
The results from the Poisson count model with robust standard errors were similar to the negative binomial model run during sensitivity analyses, whereby the rate of CRC mortality increased with increasing levels of deprivation.
Discussion
This retrospective population-based analysis of 151,050 deaths from CRC over a 10-year period in England showed that people in the most deprived areas experience a 21-28% higher CRC mortality rate than those in the least deprived areas. The difference in inequalities is greater in males (29-46%) than in females (9%-20%). The ASMR of CRC was higher in males at all the levels of deprivation compared to females. The age-specific mortality rate of CRC was higher for males than females at all ages except 0-49, where mortality rates were similar.
The main strength of our study was that data used provided comprehensive population coverage of CRC deaths in England over the 10-year period 2011-2020, so allowing calculation of robust population-based mortality rates by sex and IMD. To our knowledge, this study provides the most contemporaneous findings on the association between socioeconomic deprivation and CRC mortality in England.
Some limitations of our study should be considered in the interpretation of results. We were unable to distinguish whether differences in CRC mortality were due to individual socioeconomic status or area-level deprivation, as IMD is an area-based measure of socioeconomic deprivation, and as such the findings could be influenced by the ecological fallacy [17]. The data were insufficient to enable identification of the source of these differences as to whether they were due to differences in screening participation, stage at diagnosis, lifestyle factors such as smoking and alcohol consumption and access to treatment services.
Of interest, a randomised control trial conducted in 1989-1997 found that given equal treatment and once adjusting for the confounding effects of age, stage at diagnosis, cancer site and sex, CRC survival in England and Wales was not associated with socioeconomic status. The authors suggested that differences in survival by socioeconomic status could be due to healthcare-related factors [18].
Both survival and mortality have been used as population-health measure to describe cancer outcomes. Socioeconomic differences in CRC survival and mortality have been reported in several countries, including England, Germany and Spain [5-13,19–22]. Interpreting associations between deprivation and CRC outcomes should be approached with caution given variations in the measures of deprivation, healthcare systems and recording of cancer registry and mortality data.
A study conducted by Arik et al. in the UK using ONS mortality data for 2001-2016, investigated regional and deprivation differences in CRC mortality rate [5]. The study found that males and females in more deprived areas experienced higher CRC mortality rate and that the inequalities in mortality rate by deprivation were wider in males than in females, which are consistent with our study findings. There was increasing trend in CRC incidence up to 2010, followed by a decreasing trend in incidence for both males and females, which may be related to the introduction of the English Bowel Cancer Screening Programme in 2006 [5]. This may have led to earlier diagnosis of CRC and subsequent improvements in mortality which may have led to the overall decrease in ASMR in England.
Also, the deprivation gap in CRC mortality rates between males in the most deprived compared to the least deprived areas decreased, but did not change in females [5]. Our results show an overall decrease in ASMR between 2011 and 2020 for males and females at all levels of deprivation, which is consistent with Arik study findings. We did not find a change in the IRR by deprivation, meaning overall mortality decreased but inequalities between levels of deprivation did not change between 2011 and 2020.
A separate study conducted an analysis of English CRC mortality data for 2007-2011 showed that males in the most deprived areas had a +22.9% ASMR compared to males in the least deprived areas (24.0 in the most deprived areas vs 18.5 in the least deprived areas). Females in the most deprived areas had a +13.6% ASMR compared to females in the least deprived areas (14.0 in the most deprived areas vs 12.1 in the least deprived areas) [13]. For 2011 our study showed an ASMR of +25.2% for males in the most deprived compared to the least deprived areas and of +16% for females in the most deprived vs least deprived areas (see supplementary table 2).
The overall increase in crude CRC mortality rates with a corresponding decrease in ASMR is likely to represent an increase in the incidence of CRC due to an ageing population as well as improvements in treatments for the disease, leading to improved survival and a reduction in mortality. There is a known deprivation gradient between socioeconomic status and CRC incidence, whereby there are higher incidence rates amongst people in the most deprived groups [23].
The ONS publishes population tables by age, sex and level of deprivation which show that the most deprived areas have a relatively younger population compared to the least deprived areas[24]. This may explain why our study found that crude CRC mortality rates are lowest in the most deprived areas, but once adjusting for age, ASMR are highest in the most deprived areas.
In our study, differences in mortality rates were seen both by level deprivation and sex. Males in the most deprived areas had a +24.0% ASMR than males in the least deprived areas, and females in the most deprived areas had an +17.2% ASMR than females in the least deprived areas.
Considering that about 54% of CRC are potentially preventable and that males and females in the most deprived areas share the same living environment and access to treatment services, differences in modifiable risk factors may explain differences in incidence rates by sex.
Smoking rates and alcohol consumption are known to be higher amongst men than women and amongst the most deprived compared to the least deprived [25–27], which may explain increased CRC mortality seen in males and people living in the most deprived areas. Furthermore, there is evidence that current smoking is associated with poorer CRC prognosis compared to never smokers [28] and that co-existing comorbidity and frailty are associated with poorer prognosis in CRC patients [29].
Inequalities in age-specific CRC mortality rates by deprivation are particularly pronounced at ages 70-79 and 80-89, and particularly in males. Engagement with CRC screening at ages 60-74 is known to reduce the incidence of CRC through early detection and removal of precursor lesions, and also improve survival outcomes. An evaluation of the English Bowel Cancer Screening programme showed that screening uptake ranged from 35% in the most deprived areas to 61% in the least deprived areas [9], and that males had lower participation rates in CRC screening [30].
Despite the universal healthcare system provided by the NHS in England, there are known differences in access to treatment by region and between socioeconomic groups [31]. Previous studies have suggested that ethnicity, age at diagnosis, stage at diagnosis, delayed diagnosis and route to presentation may partially explain differences in survival and mortality rates by deprivation [31–33].
Given mortality is a function of incidence and survival, it is not possible to infer whether differences in mortality rates by deprivation and sex are due to differences in incidence rates or survival. Future studies should examine the association between incidence rates, mortality rates and differences in survival by individual and area-level socioeconomic deprivation, to disentangle the association between CRC outcomes and deprivation and to quantify the impact of individual confounders on the association between CRC mortality and deprivation. This would be with the objective of minimising a false interpretation of the association between deprivation and CRC mortality rates due to ecological fallacy.
Conclusion
This population-based analysis of the socioeconomic differences in CRC mortality in England for 2011-2020 showed that there is an association between increasing level of deprivation and CRC mortality. These inequalities particularly affect males in the most deprived areas.
The incidence of CRC is expected to increase and 54% of cases are thought to be preventable. Clinicians and public health officials should aim on decreasing the prevalence of modifiable risk factors at the population level such as smoking and alcohol, processed and red meat consumption through education and public health campaigns to reduce these lifestyle behaviours. Furthermore, there should be focus on increasing the uptake of screening in deprived areas, particularly males in deprived areas. Also, public health education initiatives for the early recognition of signs and symptoms of CRC should be prioritised to avoid any patient or primary care related delays. This is essentially important now during the aftermath of the COVID-19 pandemic, as there are known to have been delays in diagnosis and management of CRC in England [34], which have been further exacerbated by the current cost of living crisis [35]. The inequalities by deprivation and sex in CRC mortality rates may be set to change, and further work into understanding and addressing factors that might be promoting or perpetuating inequalities in CRC incidence and survival amongst the population. This may mean focusing public health policy and interventions on those in the most deprived groups, particularly males, concentrating on prevention and early diagnosis of CRC through continuing efforts with smoking cessation, education and screening programmes, as well as prioritising strategies that enable equitable access to healthcare services and consistent high-quality care.
Acknowledgements
Emma Taylor-Gallardo performed all statistical analyses and write up of this report under the supervision of Professor Eva Morris and Dr Nazrul Islam. Advice regarding coding for graphs in STATA was provided by Dr Stephanie Ross. Nick Hall for prepared the ONS data files for this project. Professor Anjum Memon provided advice regarding the structure of this report as well as direction for the findings in the context of previous literature.
Conflict of interest
The authors declare no conflict of interest.
Ethical Approval
No ethical approval was needed as use of the ONS death certification data does not require ethical approval, however those accessing ONS death certification data require ONS Accredited Researcher Status.
Emma Taylor-Gallardo completed the ONS secure researcher training and assessment to acquire ONS Accredited Researcher Status for the completion of this project.
Abbreviations
CRC: colorectal cancer; FIT: faecal immunochemical test; HNPCC: hereditary non-polyposis colorectal cancer; IMD: English Index of Multiple Deprivation; LSOA: Lower Layer Super Output Area; ONS: Office for National Statistics; ASMR: age-specific and age-standardised mortality rates; IRR: incidence rate ratio of mortality.
References
1. IARC; WHO. Age standardized (World) incidence rates, colorectal cancer, males, all ages. GLOBOCAN. 2020.
2. Arnold M, Sierra MS, Laversanne M, Soerjomataram I, Jemal A, Bray F, et al. Global patterns and trends in colorectal cancer incidence and mortality. Gut . 2017 Apr 1; 66(4):683–91. Available from: https://gut.bmj.com/content/66/4/683
3. Bowel cancer statistics | Cancer Research UK .Available from: https://www.cancerresearchuk.org/health-professional/cancer-statistics/statistics-by-cancer-type/bowel-cancer#heading-Zero
4. Rawla P, Sunkara T, Barsouk A. Epidemiology of colorectal cancer: incidence, mortality, survival, and risk factors.2022 Jul 19; Available from: https://doi.org/10.5114/pg.2018.81072
5. Arik A, Dodd E, Cairns A, Streftaris G. Socioeconomic disparities in cancer incidence and mortality in England and the impact of age-at-diagnosis on cancer mortality. PLoS One. 2021 Jul;16(7). Available from: https://pubmed.ncbi.nlm.nih.gov/34260594/
6. NCIN. Cancer by Deprivation in England 1996-2011. Cancer by Deprivation in England 1996-2011. Available from: http://www.ncin.org.uk/about_ncin/cancer_by_deprivation_in_england
7. Kanth P, Inadomi JM. Screening and prevention of colorectal cancer. BMJ. 2021 Sep;374:n1855. Available from: https://www.bmj.com/content/374/bmj.n1855
8. Bowel cancer screening: programme overview - GOV.UK.2022 Mar 17.
9. von Wagner C, Baio G, Raine R, Snowball J, Morris S, Atkin W, et al. Inequalities in participation in an organized national colorectal cancer screening programme: results from the first 2.6 million invitations in England. Int J Epidemiol. 2011 June ;40(3):712–8.
10. Ponz De Leon M, Benatti P, Borghi F, Pedroni M, Scarselli A,et al. Aetiology of colorectal cancer and relevance of monogenic inheritance. Gut. 2004 Jan 1;53(1):115–22. Available from: https://gut.bmj.com/content/53/1/115
11. The English Index of Multiple Deprivation (IMD) 2015-Guidance.
12. Wrigley H, Roderick P, George S, Smith J, Mullee M, Goddard J, et al. Inequalities in survival from colorectal cancer: a comparison of the impact of deprivation, treatment, and host factors on observed and cause specific survival. J Epidemiol Community Heal. 2003 Apr 1 ; 57(4):301–9. Available from: https://jech.bmj.com/content/57/4/301
13. Cancer by Deprivation in England 1996-2011 .2022 Jul 19. Available from: http://www.ncin.org.uk/about_ncin/cancer_by_deprivation_in_england
14. Pampel FC, Krueger PM, Denney JT. Socioeconomic Disparities in Health Behaviors. Annu Rev Sociol. 2010 ; 36:349. Available from: /pmc/articles/PMC3169799/
15. GOV.CO.UK. English indices of deprivation 2015 - GOV.UK . English Indices of Deprivation. 2015 . Available from: https://www.gov.uk/government/statistics/english-indices-of-deprivation-2015
16. Scotland PH. Standard Populations - Datasets - Scottish Health and Social Care Open Data. European Standard Population. . Available from: https://www.opendata.nhs.scot/dataset/standard-populations
17. Ingleby FC, Woods LM, Atherton IM, Baker M, Elliss-Brookes L, Belot A, et al. An investigation of cancer survival inequalities associated with individual-level socio-economic status, area-level deprivation, and contextual effects, in a cancer patient cohort in England and Wales. BMC Public Health .
18. Nur U, Rachet B, Parmar MKB, Sydes MR, Cooper N, Lepage C, et al. No socioeconomic inequalities in colorectal cancer survival within a randomised clinical trial. Br J Cancer. 2008 Dec 12 ; 99(11):1923. Available from: /pmc/articles/PMC2600684/
19. Syriopoulou E, Morris E, Finan PJ, Lambert PC, Rutherford MJ. Understanding the impact of socioeconomic differences in colorectal cancer survival: potential gain in life-years. Br J Cancer. 2019 May 28 ; 120(11):1052–8. Available from: https://pubmed.ncbi.nlm.nih.gov/31040385/
20. Jansen L, Erb C, Nennecke A, Finke I, Pritzkuleit R, Holleczek B, et al. Socioeconomic deprivation and cancer survival in a metropolitan area: An analysis of cancer registry data from Hamburg, Germany. Lancet Reg Heal Eur . 2021 May 1; Available from: https://pubmed.ncbi.nlm.nih.gov/34557810/
21. Jansen L, Behrens G, Finke I, Maier W, Gerken M, Pritzkuleit R, et al. Area-Based Socioeconomic Inequalities in Colorectal Cancer Survival in Germany: Investigation Based on Population-Based Clinical Cancer Registration. Front Oncol . 2020 May 29;10:857. Available from: /pmc/articles/PMC7326086/
22. Luque-Fernandez MA, Redondo-Sánchez D, Rodríguez-Barranco M, Chang-Chan YL, Salamanca-Fernández E, Núñez O, et al. Socioeconomic Inequalities in Colorectal Cancer Survival in Southern Spain: A Multilevel Population-Based Cohort Study. Clin Epidemiol . 2020;12:797–806. Available from: https://pubmed.ncbi.nlm.nih.gov/32801917/
23. Tweed EJ, Allardice GM, McLoone P, Morrison DS. Socio-economic inequalities in the incidence of four common cancers: a population-based registry study. Public Health . 2018 Jan 1;154:1–10. Available from: https://pubmed.ncbi.nlm.nih.gov/29128730/
24. ONS. Population by Index of Multiple Deprivation (IMD), England, 2001 to 2019 - Office for National Statistics . Population by IMD, England, 2011-2019.
25. Hiscock R, Bauld L, Amos A, Platt S. Smoking and socioeconomic status in England: the rise of the never smoker and the disadvantaged smoker. J Public Health (Bangkok) . 2012 Aug 1;34(3):390–6. Available from: https://academic.oup.com/jpubhealth/article/34/3/390/1560637
26. Wise J. UK survey confirms link between deprivation and smoking. BMJ . 2014 Mar 17;348. Available from: https://www.bmj.com/content/348/bmj.g2184
27. NHS Digital. Main Findings - NHS Digital . Health Survey for ENgland. 2019.
28. Ordóñez-Mena JM, Walter V, Schöttker B, Jenab M, O’Doherty MG, Kee F, et al. Impact of prediagnostic smoking and smoking cessation on colorectal cancer prognosis: a meta-analysis of individual patient data from cohorts within the CHANCES consortium. Ann Oncol. 2018 Feb 1;29(2):472–83.
29. Boakye D, Rillmann B, Walter V, Jansen L, Hoffmeister M, Brenner H. Impact of comorbidity and frailty on prognosis in colorectal cancer patients: A systematic review and meta-analysis. Cancer Treat Rev. 2018 Mar 1;64:30–9.
30. Mansouri D, McMillan DC, Grant Y, Crighton EM, Horgan PG. The Impact of Age, Sex and Socioeconomic Deprivation on Outcomes in a Colorectal Cancer Screening Programme. PLoS One . 2013 Jun 12.
31. Lejeune C, Sassi F, Ellis L, Godward S, Mak V, Day M, et al. Socio-economic disparities in access to treatment and their impact on colorectal cancer survival. Int J Epidemiol . 2010 Apr 21;39(3):710–7. Available from: https://pubmed.ncbi.nlm.nih.gov/20378687/
32. Nur U, Lyratzopoulos G, Rachet B, Coleman MP. The impact of age at diagnosis on socioeconomic inequalities in adult cancer survival in England. Cancer Epidemiol . 2015 Aug 1;39(4):641. Available from: /pmc/articles/PMC4542220/
33. Askari A, Nachiappan S, Currie A, Latchford A, Stebbing J, Bottle A, et al. The relationship between ethnicity, social deprivation and late presentation of colorectal cancer. Cancer Epidemiol . 2017 Apr 1 [cited 2022 Jul 19];47:88–93. Available from: https://pubmed.ncbi.nlm.nih.gov/28167416/
34. Morris EJA, Goldacre R, Spata E, Mafham M, Finan PJ, Shelton J, et al. Impact of the COVID-19 pandemic on the detection and management of colorectal cancer in England: a population-based study. Lancet Gastroenterol Hepatol . 2021 Mar 1;6(3):199–208.
35. Limb M. Cancer patients’ health is at increased risk from cost of living crisis, charity warns. BMJ .2022 Apr 29 ;377:o1103.
Received: March 03, 2025;
Accepted: March 25, 2025;
Published: March 31, 2025 .
To cite this article : Taylor-Gallardo E, Morris E, Ross S, Hall N, Memon A, Islam N. Socioeconomic Differences in Colorectal Can- cer Mortality: A Population-Based Analysis Using the Office for National Statistics Colorectal Cancer Mortality Data for England, 2011-2020. British Journal of Cancer Research. 2025; 8(2): 809- 815. doi: 10.31488/bjcr.208.
© The Author(s) 2025. This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/).
